Nanobubble Creation via Ultrasound (Acoustic Cavitation): A Technical Overview
Ultrasound, or acoustic cavitation, is a physical process that uses high-frequency sound waves to create and collapse microscopic bubbles within a liquid. This method is highly effective for generating nanobubbles, as the violent implosion of larger bubbles under the influence of acoustic energy fragments them into a vast number of smaller, stable nanobubbles. This process is widely used in various fields for its precision and ability to produce consistent results.
ChatGPT said:
Introduction
One of the most efficient physical methods for producing nanobubbles is through ultrasound-induced acoustic cavitation.
This technique uses high-frequency sound waves to generate and collapse microbubbles within a liquid, releasing energy that drives the formation of nanobubbles through fragmentation and recompression.
Ultrasound-Based Nanobubble Generation
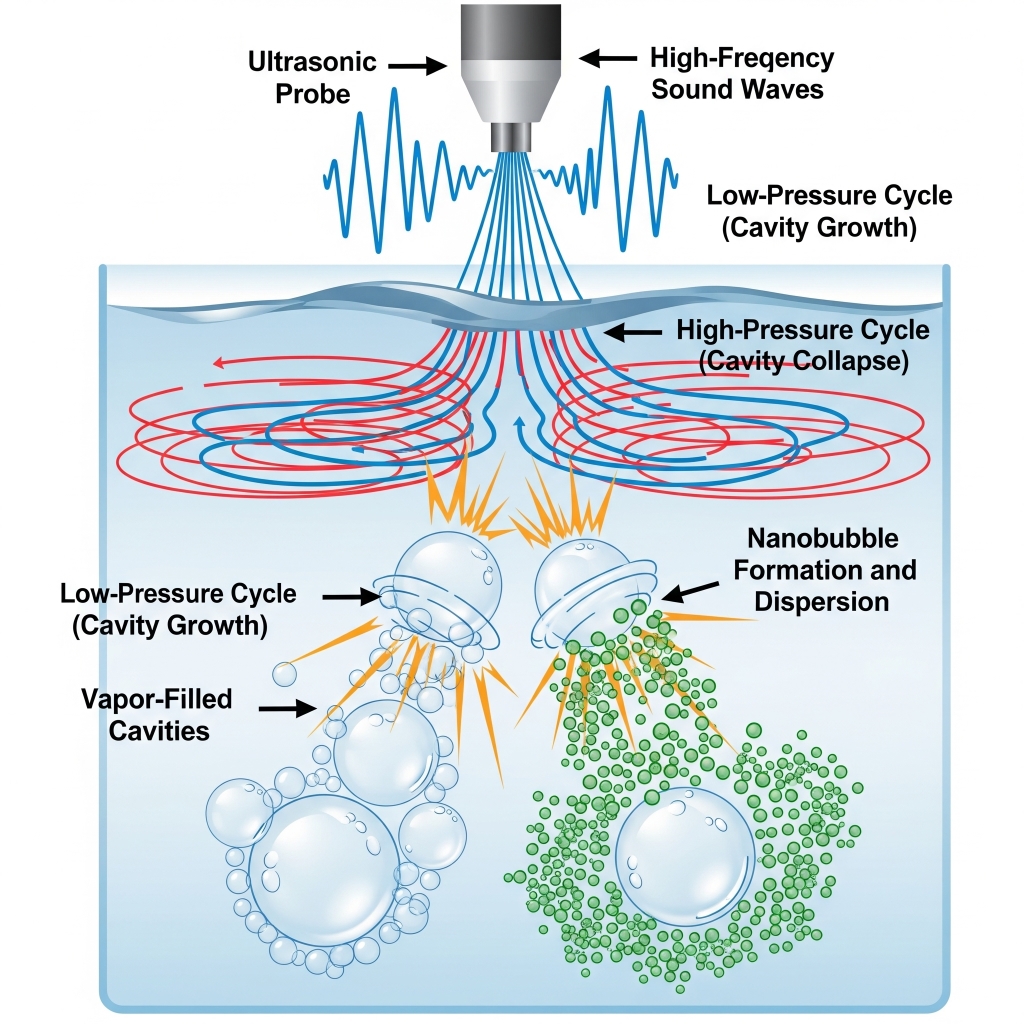
1. Introduction of Ultrasound Waves
An ultrasonic transducer (20 kHz – several MHz) is immersed in or mounted onto the side/bottom of a liquid-containing chamber. When powered, it emits high-frequency sound waves into the liquid medium.
2. Creation of Cavitation Zones
These sound waves create alternating high-pressure (compression) and low-pressure (rarefaction) cycles. During the rarefaction phase, localized pressure drops below the vapor pressure of the liquid, causing cavitation bubbles (microbubbles) to form.
3. Violent Collapse (Implosion)
In subsequent compression phases, these microbubbles collapse violently, producing localized:
- Temperatures up to 5000°C
- Pressures exceeding 1000 atm
- Shockwaves and microjets
This energy is sufficient to break up larger bubbles into nanoscale fragments or to trigger nucleation of new nanobubbles from dissolved gases.
4. Stabilization of Nanobubbles
The formed nanobubbles remain suspended in water due to:
- High zeta potential (repels coalescence)
- Near-neutral buoyancy (no rapid rise)
- Low Laplace pressure due to their small radius
They become stable and can remain in the liquid for hours to weeks, depending on the conditions.
|
Feature |
Benefit |
|
No chemicals or surfactants required |
Environmentally friendly, green process |
|
In-situ gas usage |
Air, oxygen, ozone, CO₂, or hydrogen can be used depending on application |
|
Low energy input |
Efficient with modern piezoelectric transducers |
|
High nanobubble concentration |
Generates stable bubbles uniformly dispersed in the liquid |
|
Compact and scalable |
Fits into pipelines, tanks, or reactors with modular design |
|
Versatile across gas types |
Customizable depending on oxidation or reduction goals |
Application Possibilities by Industry
Water & Wastewater Treatment
- Advanced oxidation: Ozone nanobubbles oxidize pharmaceuticals, endocrine disruptors, COD/BOD.
- DO enhancement: Oxygen nanobubbles improve aerobic digestion, nitrification.
- Biofilm control: Collapse of nanobubbles creates microjets that help in cleaning membranes and pipelines.
Agriculture & Irrigation
- Root oxygenation: Increases oxygen availability in hydroponic or flooded root zones.
- Nutrient uptake: Improves solubility and absorption of nutrients and micronutrients.
- Disease control: Ozone nanobubbles suppress pathogenic microorganisms in irrigation systems.
Aquaculture
- Oxygen delivery: Oxygen nanobubbles reduce fish stress and improve feed conversion.
- Water quality: Helps maintain higher dissolved oxygen (DO) levels and lower ammonia.
Food and Beverage
- Sterile rinsing: Ozone nanobubbles provide surface sterilization for produce and packaging.
- Emulsification and mixing: Improves homogeneity in beverages or sauces using bubble-induced turbulence.
Clean Hydrogen Applications
- Water splitting efficiency: Nanobubbles (e.g., H₂ and O₂) help improve gas separation during electrolysis.
- Electrode cleaning: Implosive force of bubbles cleans electrode surfaces, enhancing catalytic performance.
Surface Cleaning & Precision Electronics
- Ultrasonic nanobubbles dislodge particles from surfaces and blind holes in semiconductors and optical components.
- Used in combination with DI water or mild solvents to reduce chemical load.