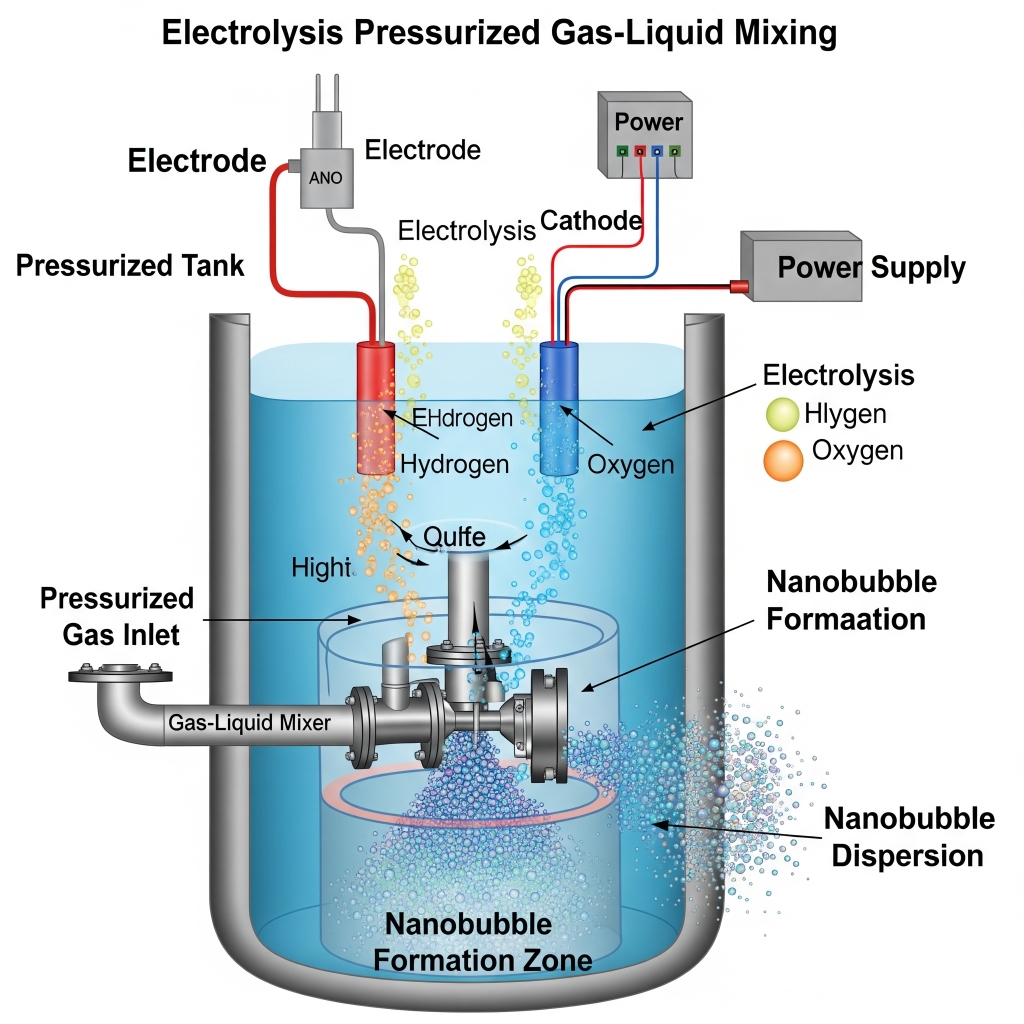
How are Nanobubbles Created Using Pressurized Gas–Liquid Mixing
Pressurized Gas–Liquid Mixing is essentially a mass-transfer acceleration process engineered to force gas into water under controlled pressure, then rapidly release that pressure to trigger nanobubble nucleation. It’s predictable, scalable, and easy to integrate into industrial infrastructure.
Think of it as a closed-loop gas infusion system that takes advantage of supersaturation physics.
A Simplified Explanation
Nanobubbles are created by pushing gas into water under pressure, then releasing it instantly. In a pressurized mixing chamber, water and gas are forced together until the water becomes supersaturated; meaning it’s holding more gas than it normally can. When this high-pressure mixture is released back to normal pressure, the dissolved gas rapidly forms billions of nanobubbles (typically 80-nm to 200-nm).
These nanobubbles stay suspended in the water because they are too small to rise, and they carry a natural negative charge that prevents them from merging together. The result is a long-lasting, highly reactive, oxygen-rich water environment that improves oxidation, boosts treatment efficiency, and enhances water quality across wastewater, aquaculture, agriculture, and premium swimming pool applications.
This is why pressurized gas–liquid mixing remains one of the most scalable and reliable nanobubble generation methods for industrial and municipal operations.
Pressurized Gas–Liquid Mixing: The Process in Detail
Process Overview
- Gas (e.g., air, O₂, O₃) is dissolved into a liquid (typically water) under high pressure.
- The gas–liquid mixture is intensively sheared using a static mixer, venturi injector, or pressurized chamber to create micro/nano-sized bubbles.
- The supersaturated solution is then rapidly depressurized, which causes nanobubbles to nucleate and form as the gas exits solution.
Core Mechanism
- Henry’s Law governs how much gas can be dissolved into the liquid under pressure.
- When pressure is released, dissolved gas wants to come out of solution.
- Due to high shear and turbulent conditions, instead of forming large bubbles, nanoscopic gas nuclei form and remain stable due to surface charge (zeta potential) and lack of buoyant rise.
Typical Setup
- High-pressure pump
- Gas injector (pressurized gas line)
- Static mixer or shear chamber
- Pressure-release valve or nozzle
Pros
- Energy-efficient for many applications
- Scalable and relatively low maintenance
- Works well with various gases (O₂, O₃, CO₂)
Cons
- Requires precise pressure controls
- Bubble size distribution may be broader without tuning