Nanobubble Generation via Electrolysis (Electrochemical Method)
Process Overview
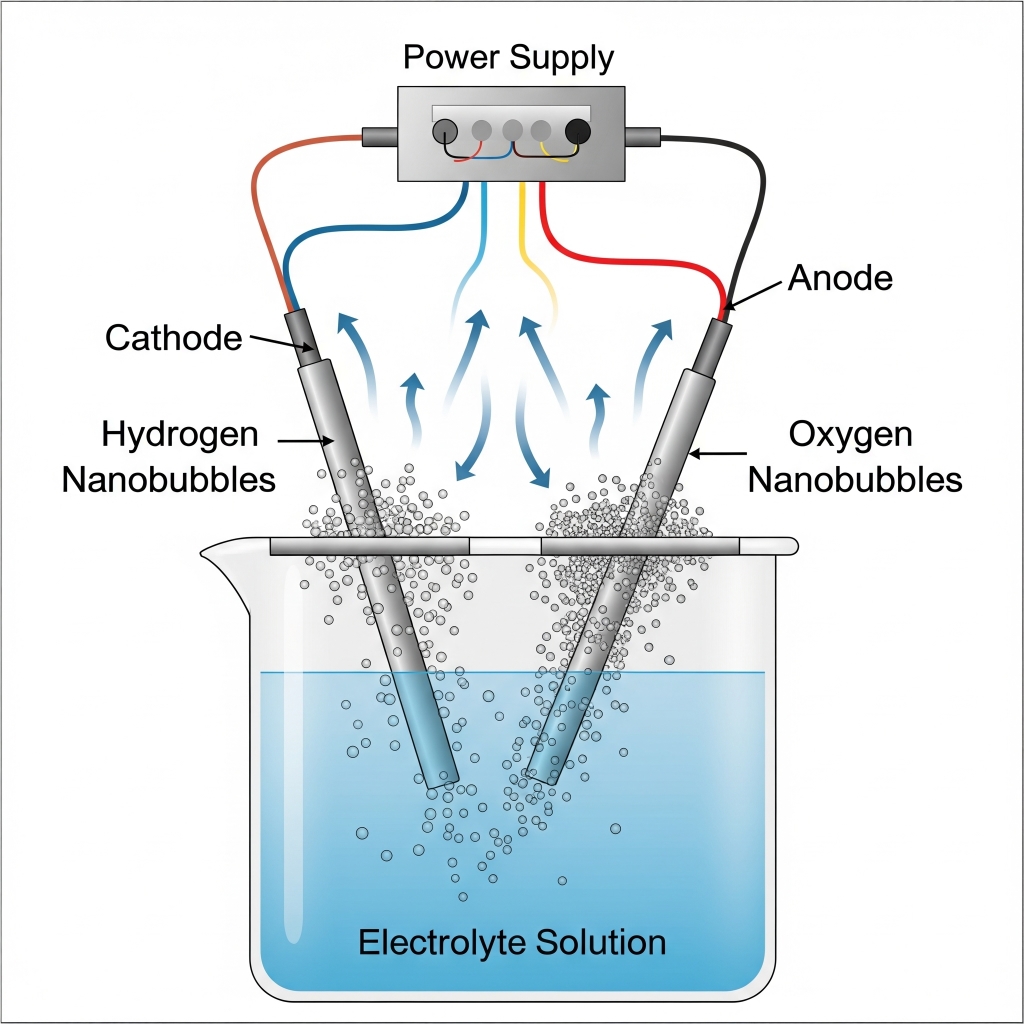
Electrochemical nanobubble generation involves using electric current to split water molecules (H₂O) into hydrogen (H₂) and oxygen (O₂) gases at two electrodes submerged in water. Under specific operating conditions, these gases do not coalesce into large bubbles but instead form ultra-fine gas nanobubbles (<200 nm in diameter) that remain suspended in water.
Electrolysis, The Process in Detail
System Setup
A power supply is connected to two electrodes:
- Anode (+): Oxidation occurs (O₂ evolution)
- Cathode (−): Reduction occurs (H₂ evolution)
Electrodes are submerged in a clean water-based electrolyte (e.g., Na₂SO₄ solution or tap water with a conductive salt).
Electrochemical Reaction
When current is applied:
At the Anode (+): Oxygen gas forms at the surface.
At the Cathode (−): Hydrogen gas forms at the surface.
Nanobubble Formation
Supersaturation Zone: Near the electrode, the local concentration of dissolved gases exceeds solubility, creating high gas pressure in a very small volume.
Nucleation: Instead of forming large bubbles that float upward, tiny gas nuclei form due to interfacial tension and electric field effects.
Stabilization: These nuclei grow to nanobubble size and become electrostatically stabilized by surface charges (zeta potential), preventing coalescence.
Dispersal: The nanobubbles are released into the surrounding liquid and remain suspended for hours or even days due to their neutral buoyancy and Brownian motion.
Key Benefits
Benefits of Electrochemical Nanobubble Generation
|
Feature |
Benefits |
|
No Moving Parts |
A simple and compact, low-maintenance system |
|
On-Demand Production |
Generates nanobubbles at point of use without gas storage |
|
High Purity |
Produces ultrapure H₂ and O₂ nanobubbles, ideal for sensitive processes |
|
Scalable |
Easy to adapt from lab-scale to full industrial-scale systems |
|
Chemical-Free |
No need for external surfactants or stabilizers |
|
Selective Gas Injection |
Enables independent or combined O₂/H₂ delivery |
Industrial Applications
1. Clean Hydrogen Production: Used in alkaline or PEM electrolyzers where nanobubbles improve electrode efficiency and reduce overpotential. Enhances gas separation and reduces recombination losses.
2. Wastewater Treatment: Electrochemically generated oxygen nanobubbles boost aerobic biological processes and oxidation of pollutants. Hydrogen nanobubbles support advanced reduction processes (ARPs).
3. Semiconductor & Ultrapure Water: Used to remove submicron particles from wafers and prevent surface oxidation.
4. Agriculture and Hydroponics: Nanobubbles enhance root oxygenation and reduce pathogens in irrigation water.
5. Food & Beverage: Improves sanitization and shelf life during washing, mixing, or fermentation processes.
6. Cooling Towers & Boilers: Oxygen nanobubbles control biofilm and improve heat transfer while reducing chemical load.